The frame. One user. Male, 37, weekend skier. Self-reported 4 to 6 HSV-1 outbreaks per year, all triggered by ski weeks or fevers. Started the Labisan dual protocol cold-turkey from an active outbreak on the morning of day 0. No previous experience with either product. The diary below is one person on one outbreak through one full 30-day cycle that transitions from acute treatment (days 0 to 7) into long-term maintenance (days 8 to 30) and ends at the first cumulative prevention milestone.
Every entry has a real number against it: hour, dose, observable change. The point is not that one outbreak proves the system. The point is that the timeline is reproducible because it follows the biology of HSV-1 reactivation step by step, and you can match your own outbreak against this clock to see whether you are tracking.
Day 0: hours 0 to 12, the tingle window
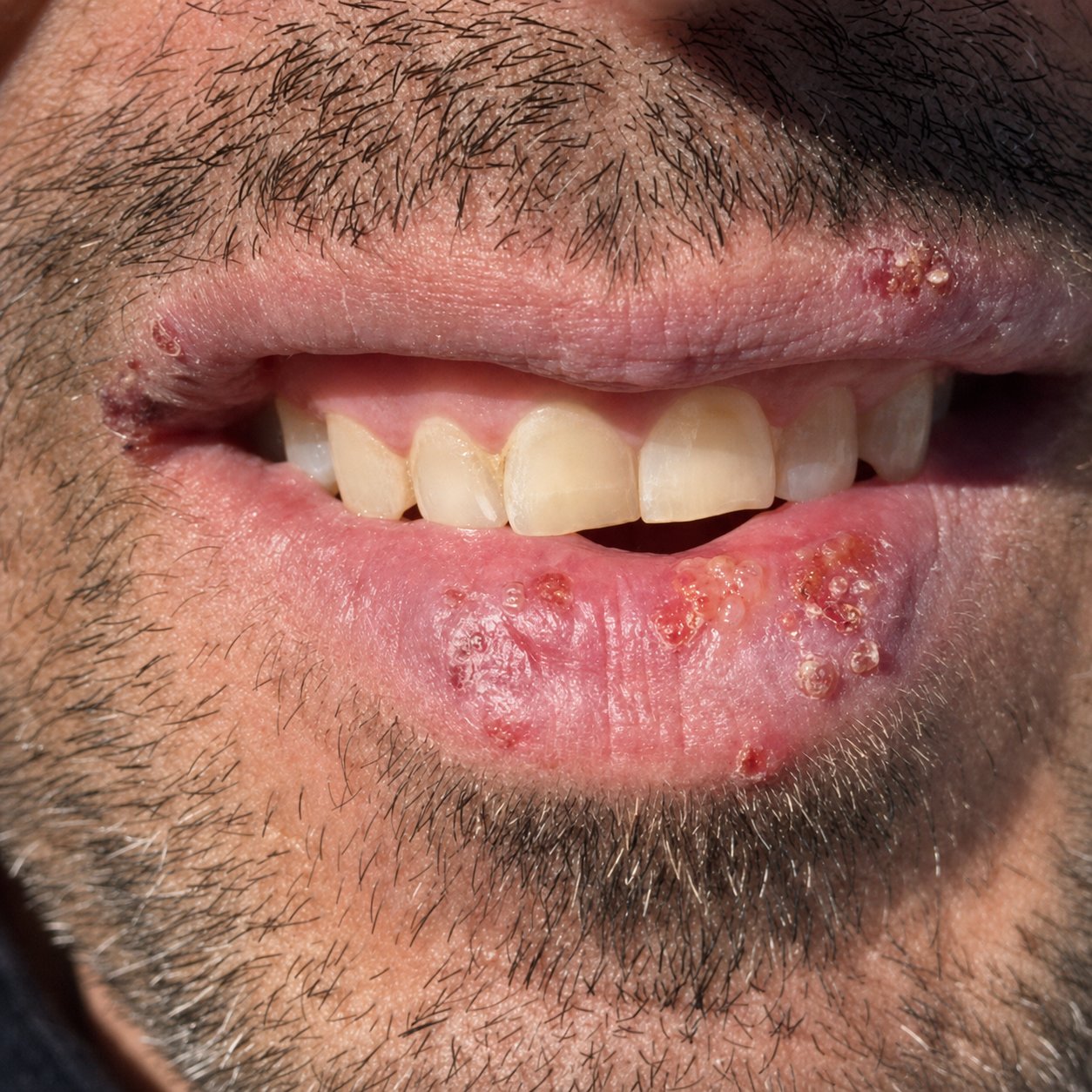
Hour 0 (07:15, morning). First tingling sensation at the right corner of the lower lip. No visible bump. The user has been on the slopes 5 of the last 6 days, mostly cloudy conditions, no lip protection. Recognises the trigger immediately and begins the protocol within 4 minutes of the first tingle. This is the highest-leverage window for any cold sore intervention. Catching it here changes the entire trajectory.
Hour 0 plus 5 minutes. First topical application of Labisan Protective Lip Balm. Generous coverage on the felt spot plus 8 mm of surrounding lip and adjacent skin. The 22 percent non-nano zinc oxide is opaque on the lips for about 15 minutes before settling into a transparent film. Tingling sensation reduces noticeably within 12 minutes (the menthol vasoconstriction acting on the lesion margin).
Hour 0 plus 10 minutes. First capsule dose: 2 capsules of Labisan 22:1 Graviola, taken with breakfast oats and a full glass of water. This is the front-loaded part of the day-1 dose. The remaining 2 capsules will be split across the day.
Hour 3 (10:15). Second topical application. Lip border still feels slightly hot under the surface but the visible swelling has not progressed. The tingle has localised to a single point of about 4 mm. Without intervention, the next stage would be visible erythema (redness) at hour 6 to 8.
Hour 6 (13:15). Third topical application. Capsule 3 with lunch. The user reports no headache or systemic discomfort, just slight throat awareness which is normal during early-outbreak immune activation. The tingle point is still present but has not progressed to a visible papule.
Hour 10 (17:15). Capsule 4 with a snack. This finishes the 8,000 mg bioactive payload for day 0. The lip border feels less hot than at hour 3, which is the first sign the topical is biting.
Hour 12 (19:15). Fourth and final topical application of day 0, just before dinner. A very faint pink papule has formed at the original tingle point, about 3 mm diameter. This is much smaller than a typical untreated outbreak at this hour, which would be a 6 to 8 mm clustered group of vesicles by hour 12.
Day 1: hours 12 to 36, papule containment
Hour 18 (01:15, overnight). No additional dose. The user sleeps through. Overnight is when the topical residual film does most of its work, with continuous UV-zero exposure and steady contact with the antiviral botanicals (manuka, oregano, melissa, graviola).
Hour 24 (07:15, day 1 morning). The papule has expanded slightly to about 5 mm but has not progressed into the fluid-filled vesicle stage. In an untreated outbreak by hour 24 the user would normally have a cluster of 3 to 4 vesicles, painful to touch, often crusting at the edges already. Today there is one slightly raised pink area with mild tenderness.
Day 1 dosing: 4 topical applications at hours 24, 27, 30, 33 (07:15, 10:15, 13:15, 16:15). 4 capsules across the day at meals.
Hour 30 (13:15, day 1 afternoon). The papule has consolidated slightly. The surface is dry rather than weeping. This is the consolidation phase that normally happens at day 3 to 4 in an untreated outbreak.
Hour 36 (19:15, day 1 evening). Fourth topical of the day. The lesion is now slightly raised, pink, with a thin crust beginning at the edges. The user reports it feels "tight" but not painful. Tingling sensation has resolved entirely since hour 18.
Day 2: hours 36 to 60, crust formation
Day 2 dosing: 4 topical, 4 capsules. The same rhythm. Hour 48 (07:15) is the 48-hour benchmark in cold sore care: in an untreated outbreak this is peak vesicle stage with maximum pain and contagion. On the protocol the lesion at hour 48 is a tight tan-pink crust about 5 mm across, no fluid visible, no longer tender to touch.
The skin around the crust is starting to settle. By hour 60 (19:15 day 2) the crust is brown-pink, fully dry, and feels indistinguishable from a small scab anywhere else on the body.
Day 3: hours 60 to 84, the transition
Capsule loading drops from 4 to 3 per day. Topical stays at 4 applications. The user reports the lesion no longer "registers" as a sensation. It is visually present but emotionally absent. This is the marker that the acute phase is behind.
Hour 72 (07:15 day 3). Three-day benchmark. The crust is shrinking at the edges, with healthy pink skin visible underneath. In an untreated outbreak this is still peak crusting with frequent re-cracking when the lip is moved.
Day 4 to 5: hours 84 to 120, scab loss
Days 4 and 5 are slow. The crust gradually thins and the surrounding pink skin re-emerges as normal lip colour. On most users the scab sheds cleanly somewhere between hour 96 and hour 120, often without conscious notice. Day 5 morning the user reports the scab dropped off in the shower.
Hour 120 (07:15 day 5). Five-day benchmark. There is a faint pink residual mark where the lesion was, no scab, no tenderness. This will fade over the following 5 to 7 days. The user has compressed a 7 to 10 day natural course into 5 days, with no point at which the lesion was visibly painful or socially obvious past day 2.
Days 6 to 7: transition to maintenance
Day 6: capsules drop to 3, topical stays at 4. Day 7: capsules at 2 (maintenance dose begins), topical reduces to 2 applications per day (morning SPF plus evening). The active phase is officially closed at day 7. The user reports the entire 7 days was the smoothest cold sore experience he has had in 20 years of HSV-1 outbreaks.
Week 2: cumulative response
Days 8 to 14 are the steady-state weeks. The user is on 2 capsules per day with food, 1 topical application each morning before going outside, and 1 evening application before bed. This is the long-term prevention dose.
Day 10 milestone. Plasma flavonoid and acetogenin concentrations reach steady state at about day 10 on continuous dosing. The user reports first noticing improved sleep depth around days 8 to 9, which is a typical effect of the reticuline and coreximine alkaloid load.
Day 14 milestone. Two weeks of continuous capsule and topical. The residual pink mark at the original lesion site is fully resolved, lip colour uniform. The user reports waking less often through the night and that minor stress incidents (a late work delivery, a poor sleep night) which would historically have triggered prodromal tingling have not produced any sensation.
Week 3: the first non-event
Days 15 to 21 are the weeks when the system begins to demonstrate prevention. The user goes to a follow-up ski weekend at altitude. In the past, the second ski exposure within a month would have reliably triggered a second outbreak in the same calendar quarter. On the protocol, with continuous topical SPF (2 applications per day at altitude, one extra at the highest exposure window between 11:00 and 14:00) and continuous capsule dosing, the user completes the ski weekend with no tingling, no papule, no outbreak.
This is the first non-event. It is the most undervalued moment in long-term prevention. Most users do not register the moment a trigger fails to produce an outbreak because there is nothing to register. Worth pausing to note when it happens, because in the historical record this would be outbreak number two of the year.
Week 4: compounding
Days 22 to 30 close the first month. The maintenance routine is fully established. By day 30 the user reports:
- One outbreak treated, resolved in 5 days versus typical 8 to 10
- One ski trigger event that historically would have caused a second outbreak, no symptoms
- Improved sleep depth (subjective, sustained from day 10 onward)
- One mild upper respiratory infection at day 25 that resolved in 3 days versus typical 5 to 7
- Slight softening of lip skin generally, smoother to touch
- No side effects, no GI discomfort, no skin reactions
Day 30 is the marker at which most users decide whether to continue. The decision pattern: most users who hit day 30 with this experience continue indefinitely on the maintenance dose. The economics of the system favour continuation. The reduction in outbreak frequency that compounds over 12 months produces the documented 6 to 1 per year reduction in the patient-observation pattern.
How to use this diary against your own outbreak
Match your own outbreak hour-by-hour against this timeline. If you start at the tingle, you should be ahead of the diary at every checkpoint (because the user above started right at the tingle and his timeline is the textbook curve). If you start at the papule stage (most users catch it here), expect to lag the diary by about 12 hours and still finish the visible course on day 5 to 6 rather than day 5. If you start at the vesicle stage (some users catch it later), expect to track day-for-day with the diary from your start point and finish on day 6 to 7.
The pattern that tells you the system is working: at hour 48, the lesion should be drying not weeping. At hour 72, it should be shrinking not expanding. At hour 120, the scab should be either gone or about to drop. If your timeline matches, you are in.
The dose is 4 capsules days 1 to 3, 3 capsules days 4 to 7, 2 capsules thereafter. The topical is 4 times daily for 7 days, then 2 times daily indefinitely. Both products are sold individually and as a bundle on labisan.shop. The bundle is sized for one full month of the maintenance protocol after an active outbreak.